|
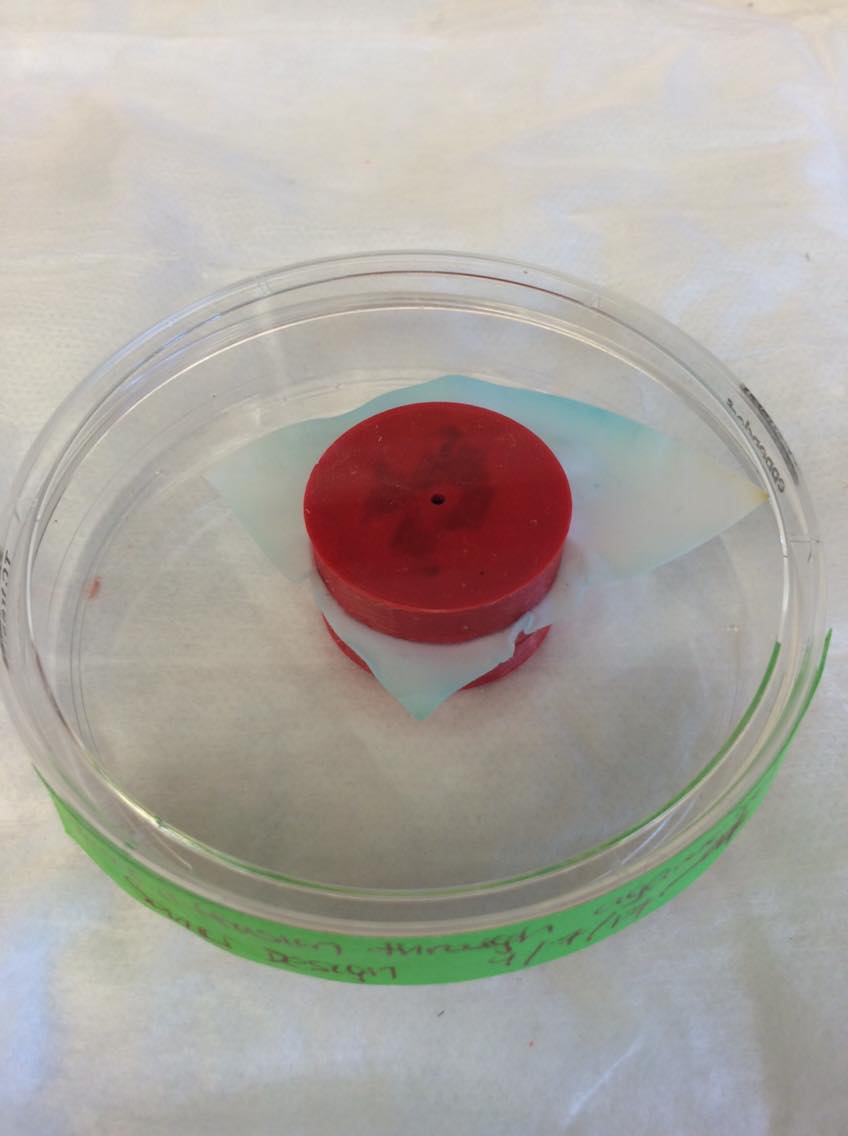
-For this week, our group focused exclusively on testing of the prototype device with a .5% agarose gel that had no channel puncture through it. Specifically for the purposes of this experiment, we were looking to identify four things: 1. How would the gel loading procedure look like within the closed chamber device when the gel was loaded onto the filter paper surface via a pipet tip that fit into the top cap. 2. Would the filter paper hold the gel? 3. Would dye from the top of the gel diffuse through it and reach the bottom of the gel 4. Would the entire structure remain intact over a long period of time To examine these things, we prepared the gel and loaded it within the device. During the first run through, the filter paper was punctured by the channel creation process which led us to increase the length of the overlap in the final design. In a second run through, we were able to successfully ensure that the gel would remain in the top compartment and after waiting 10 mins for it to solidify, we loaded a dye via the drug injection port and came back the next day to check on it. We were able to see very good results and they are shown in the pictures attached below. For the next step, we have gone ahead with printing our final design using finer material and plan to create a channel that will have liquid being pumped through it for an extended period of time. Afterwards, we plan to load Rhodamine B to the top and see if we are able to collect any of that from the channel flow through. This signal can be read via a fluorometer. The attached pictures show the gel loading process, that the dye was able to diffuse uniformly from the top of the gel all the way to the very bottom, what it looks like while the experiment was run, and hat the gel remains confined to the top of the device (above the filter paper) for the whole time. -Arnold
0 Comments
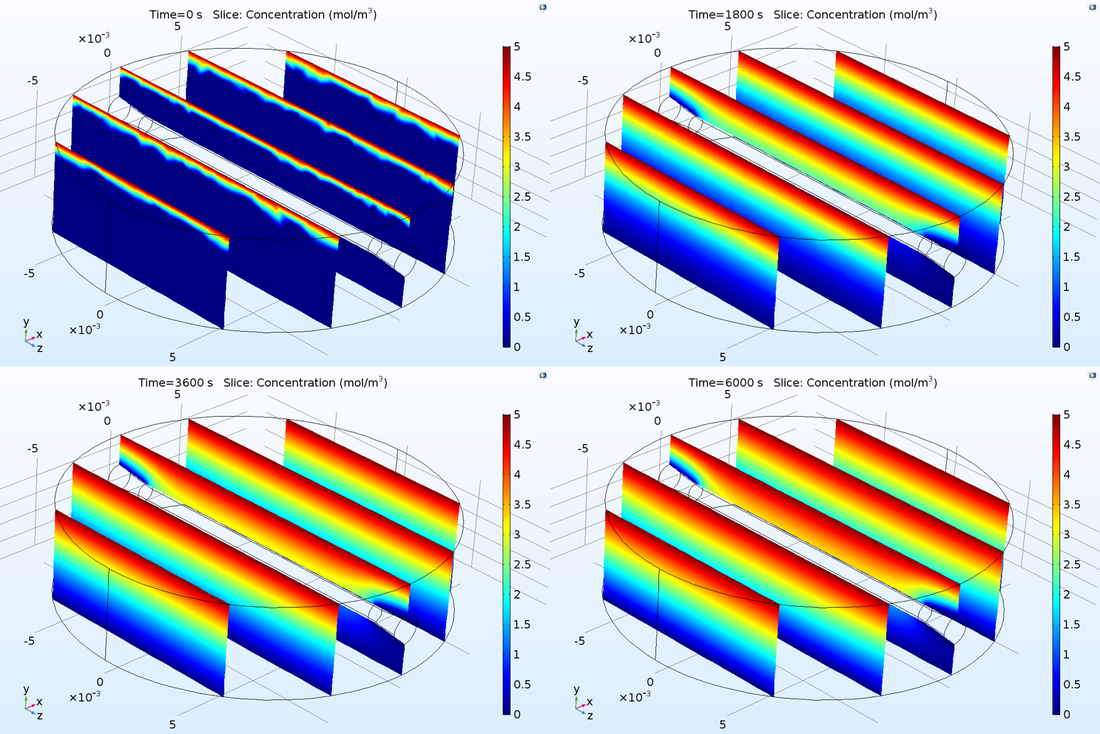
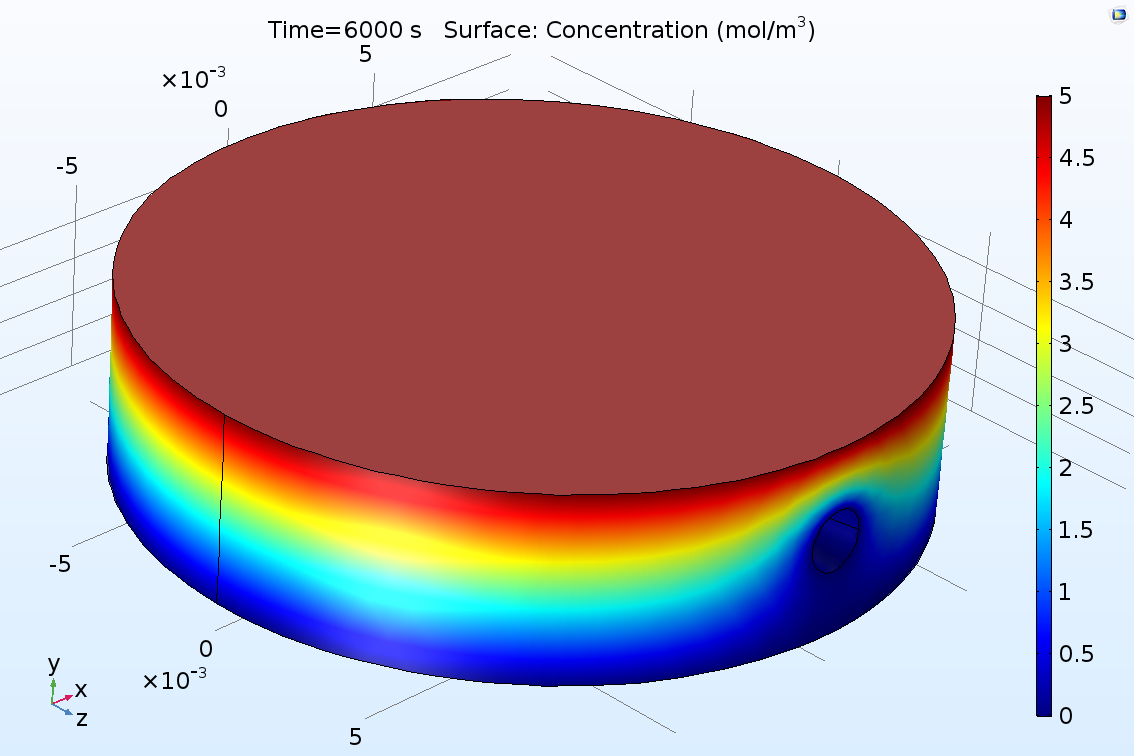
In the past week, we received 3D printed modules of the final design of drug chamber. Moreover, 19 gauge needles for channel creation and 1/16in connectors that we ordered were delivered during the week, which will allow us to validate the interface between the chamber and the fluid delivery/collection system. Our group met with our client's representative Young on Wednesday morning and we as a team decided to meet again for loading test on Friday which would happened after this report is written so detailed results shall be recorded in next week's report. During this week we also were working on improving the simulation model in COMSOL for diffusion and more testing on channel-in-gel integrity maintenance. Attached are our unanalyzed results. Further analysis shall be done during this weekend.
-Richard Dear Dr. Yin and Ali,
We are in the process of printing an ABS prototype of our cylindrical design to test the feasibility of our lid design to prevent leakage. The ABS prototype will give us a better idea of what parameters need to be adjusted to ensure the best fit between the three compartments. 19 gauge needles for channel creation and 1/16in connectors have been ordered and are on track for delivery next week, which will allow us to validate the interface between the chamber and the fluid delivery/collection system. Best, David Sun Thank you again for meeting with us last Monday. Our discussion was constructive and allowed us to focus on short term objectives to meet expectations for the V/V report as well as elucidate long term expectations for the prototype. We have since communicated the notes of the meeting to our client representative.
Since then, the group has been focused on finalizing our design in CAD and modeling flow through the chamber in COMSOL. The group will be meeting later today to collaborate on the design and model and allocate work for the upcoming V/V report and presentation. I will be focused on refining the chamber design through changes which include an interlocking lid design to prevent inter-compartmental leakage between the chamber segments. Richard and Arnold will carry the responsibility of creating the COMSOL model. Best, David Sun During the brief meeting this week, the alternative cylindrical design that was discussed in last meeting was published in CAD and its dimensions were discussed in this week’s meeting as well.
Regarding the unibody design, we ran some experiments to improve the integrity of channel embedded in the gel to achieve the goal to maintain an open conduit for pressure driven flow. The measures that were taken during the experiments include increasing the channel size (radius), incubating the loaded gel (within testing chamber) under higher temperature and so on. Lastly, out group also discussed the future plan that to merge the features of two designs we are currently testing into one. Namely, after the improvement of conduit fabrication in the unibody design, the procedure shall be applied to the cylindrical design to improve its stability for diffusion measurement performance. In the end, attached are the top, middle and bottom compartment of the new cylindrical design. The top piece shall be holding the initial load of drug solution, the middle compartment shall be holding the gel (agarose, matrigel, etc.), the bottom piece shall serve the purpose of end fluid collection. More details of the design will be brought into next week’s report after more group discussions and experiments. Firstly, apologies for the tardy progress report. Our meeting with our client just ended (12:00PM).
In the meeting, we have discussed more alternatives to the design. We plan to revisit the cylindrical chamber design at the request of the client and plan to publish a CAD model by this weekend. Another point of discussion was the need for our group to seek out an expert. Because the point person on the project is no longer part of the lab, we agreed to begin seeking advice from experts in the field. Our group also plans on conducting another experiment with our prototype with stiffer agarose gel, something that was originally scheduled last semester but was not completed due to conflicts. Based on feedback from you on the progress report and presentation as well as advice from our client, we plan on editing our specific aims to better satisfy the requirements of the course. Most of our time this week was dedicated to the progress report and presentation next week. During the compilation of this report, we considered a lot of different methods and have most solidified the ones we find most promising. This helped us to clarify our future direction and we now have a better idea of what components still need more work moving forward. We also met with our client this morning to update him on our project. We plan to change the chamber design and test with a stiffer design using the needle method. We hope to have this done before the semester ends.
Last Friday afternoon (11/18), the team met up to test a 300 um-diameter needle coated in BSA for channel formation. We first made up 0.5% agarose and loaded it into the chamber with the needle inside. After the gel solidified, the needle was removed carefully using tweezers. We first tried pushing fluid through with a 20 uL pipette. The fluid chose to pass through the space where the gel met the chamber wall instead of through the channel itself. This led us to believe that 300 um may be too small and confer too much resistance to allow fluid to pass. In addition, we were not able to see if the needle removal left a channel that was able to support itself due to the very small diameter. In future experiments, we will test larger diameter needles and use a dye to track fluid flow through the channel.
On 11/22, I met with Nick Thompson to discuss modifications to the chamber design. We discussed printing a channel out of PLGA or PCL to solve the channel instability issue. However, there were significant limitations with the low permeability and cost of such a channel. We also discussed the possibility of a needle with small enough perforations to inhibit the PEG to flow into the chamber. Pictures and video from our 11/18 experiment are uploaded to the following folder: https://drive.google.com/drive/folders/0Bz1SvoM7LVxSQ09FNlBrLWZVcDQ?usp=sharing At the meeting last Friday afternoon with our client on 11/11, we discussed the material properties of our gel chamber and decided to look further into whether or not the materials we wanted to 3D print with would absorb small molecules like urea which is 60 daltons (very small). We also discussed methods to sterilize the device as the autoclave is not an option for most 3D print materials. The high temperatures and pressure that that the autoclave goes up to will end up deforming most 3D print materials that we use.
In our meeting, we also discussed other options to look into. Due to design and print difficulties, our group has decided to try out needles using agarose gel as a proof of concept. From speaking to other researchers in the George Lab who have expertise in using needles that interact with ECM and PDMS gel interfaces, we have decided to submerge our needles in bovine serium albumin (BSA) for 30 minutes in an attempt to decrease the friction coefficient and make it much easier to remove from the gel. An official protocol will be uploaded next week after further discussions with George Lab members. We have also asked for scientific literature and protocols from the lab, which we are in the process of getting. Further, we will be testing the BSA needles in an agarose gel in order to assess the plausibility of this strategy. The needle will be inserted into agarose gels at varying concentrations, allowed to polymerize, and then removed to assess any damage that is done to the gel afterwards. Protocols will be uploaded as these experimental designs are finalized. We are excited to start testing out different components in order to assess the feasibility of our ideas. At the same time, we are also developing back-up plans in case aspects of this prove to be beyond our capabilities. |
Group 38Archives
April 2017
Categories |